
A series of effective close-packing volumes for a range of ions, which will be of interest to chemists, as measures of relative ionic size and which are of use in making our estimates of lattice energies, is generated from our approach. Type of enthalpy change, Value of enthalpy change/ kJ. Thus +2 or -2 ions will release more lattice energy than the +1 or -1 ions. where b Repulsion coefficient or proportionality constant or Born coefficient. of the free energy associated with the lattice vibrations in an anharmonic. Lattice energy increases with increase of charge on the ions because of their more attractive force between them. Theoretical calculation of lattice energy . B7, EQUATION OF STATE AND THERMODYNAMIC FUNCTIONS OF NON-METALLIC CRYSTALS. This is illustrated by accounting for the failure to prepare diiodinetetrachloroaluminum(III), and the instability of triiodinetetrafluoroarsenic(III). The table shows the enthalpy changes needed to calculate the lattice energy of potassium oxide, K 2 O. Thus the attraction between them decreases and finally the less lattice energy released during the process. As is demonstrated, the approach can be utilized to predict and rationalize the thermochemistry in topical areas of synthetic inorganic chemistry as well as in emerging areas. However, our new equation can be employed even when the latter information is not available. The Born-Land equation was derived from these two following equations. Going across the periodic table, atomic radii decreases, therefore lattice energy increases. Element X reacts with sodium to form an ionic compound with the formula Na2X. Lattice energy decreases as you go down a group (as atomic radii goes up, lattice energy goes down). 
In such cases, lack of information about cation-anion distances prevents use of the Kapustinskii equation to predict the lattice energy of the salt. Because of their smaller size and higher charge, the hydration energies of. 
When new salts are synthesized, acquisition of full crystal structure data is not always possible and powder data provides only minimal structural information-unit cell parameters and the number of molecules per cell. We have generalized Bartlett's correlation for MX (1:1) salts, between the lattice enthalpy and the inverse cube root of the molecular (formula unit) volume, such as to render it applicable across an extended range of ionic salts for the estimation of lattice potential energies. Each unit cell is defined in terms of lattice points the points in space about which the particles are free to vibrate. It is defined as the energy required by a body to accelerate from rest to stated velocity. U, which is always a positive number, represents the amount of energy required to dissociate 1 mol of an ionic solid into the. We will sum the energies of each step and that will be our total. Answer: UL 755 KJmol Example 2: The lattice energy of. We will use Hesss Law to rearrange the steps so that we get the net equation we are after. The science, which deals with crystals properties is. The lattice energy of nearly any ionic solid can be calculated rather accurately using a modified form of Equation 4.2.1: U kQ1Q2 r0, where U > 0 (4.2.1) (4.2.1) U k Q 1 Q 2 r 0, w h e r e U > 0. Lattice Energy Formula Example 1: Compute the Lattice energy of NaCl by using Born-Lande equation. 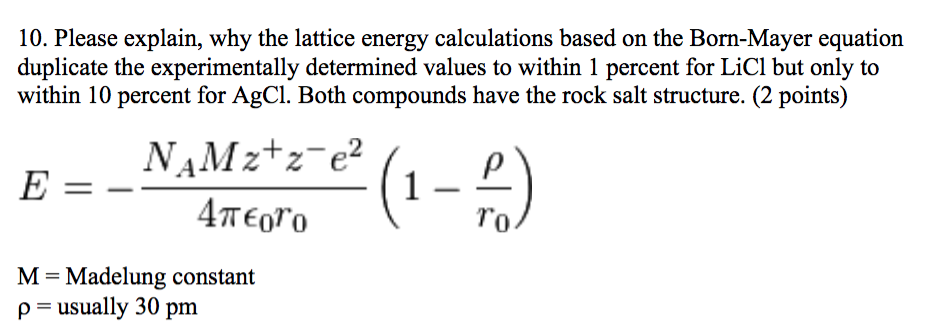
The lattice energy is usually given in kilojules per mole (kJ/mol). The lattice energy is the total potential energy of the crystal. Second, it makes possible the acquisition of lattice energy estimates for salts which, up until now, except for simple 1:1 salts, could not be considered because of lack of crystal structure data. The Coulombic equation is a commonly used formula for calculating the lattice energy of an ionic compound. The kinetic energy of an object is the energy associated with the object which is under motion. Equivalently, lattice energy can be defined as the amount of work (energy) that is released during creation of crystal lattice from ions separated to infinity. First, it offers an alternative (and often more direct) approach to the well-established Kapustinskii equation (whose capabilities have also recently been extended by our recent provision of an extended set of thermochemical radii). \( \newcommand\)).The linear generalized equation described in this paper provides a further dimension to the prediction of lattice potential energies/enthalpies of ionic solids.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
